David Savastano, Editor04.25.12
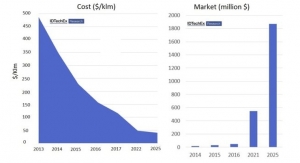
The ability to print or mass produce electronics, preferably on roll-to-roll systems, is the promise behind printed electronics (PE). Not only could a company produce their products at a much less expensive cost, but printing on flexible substrates opens the doors for many new applications.
While a few products such as organic light-emitting diodes (OLEDs) and organic photovoltaics (OPVs) are already being printed, there are plenty of opportunities awaiting solutions. For example, large solar cells and displays could ultimately be printed at a fraction of the costs of today’s systems. However, there are technological hurdles that need to be solved.
One of the key hurdles is utilizing metals in ambient conditions. Conductors such as calcium, magnesium or lithium oxidize when exposed to moisture and oxygen. This requires encapsulation or glass to protect these metals. If a protective polymer can be developed that can be printed in a thin layer, that would go a long way toward solving this issue.
Researchers at Georgia Tech’s Center for Organic Photonics and Electronics (COPE) believe they may have found the answer to this problem. In new findings published in the journal Science, Georgia Tech researchers spread a very thin layer of a commercially available, environmentally friendly polymer, approximately one to 10 nanometers thick, on the conductor’s surface to create a strong surface dipole. The interaction turns air-stable conductors into efficient, low-work function electrodes.
Prof. Bernard Kippelen, director of Georgia Tech’s Center for Organic Photonics and Electronics (COPE), said that the discovery of these stable materials was a result of COPE’s research expertise in numerous fields.
“As often the case in science, the first lead that enabled this discovery was the serendipitous observation that a polymer dispersant that was initially used to help disperse metal oxide nanoparticles was producing unexpected changes of the work function of indium tin oxide electrodes,” Prof. Kippelen said. “A closer look at the chemical structure of this non-conjugated polymer brought us to believe that amine-containing polymers could have some interesting electronic interactions with electrodes that could lead to a reduction of their work function.”
Prof. Kippelen and his team took their findings to their colleagues in Georgia Tech’s Chemistry and Physics departments, who see much potential.
“Sharing of that observation with our closest collaborators in chemistry led to the selection of a few commercially available branched polymers containing simple aliphatic amine groups, including branched polyethyleneimine (PEI) polymers,” said Prof. Kippelen. “The testing of these materials revealed significant reduction of the work function of a variety of conductors. A series of experiments and theoretical modeling with additional collaborators allowed us to get a good physical understanding of the process.
“The validation of the reduction in work function of electrodes in various organic optoelectronic devices exceeded our expectations,” Prof. Kippelen added. “In particular, the good performance of these stable polymers in organic solar cells made us believe that we had made an important discovery that could have a game-changing impact on printed electronics.”
The polymer works with a wide range of metal conductors, including silver, gold and aluminum, as well as graphene and metal oxides. To showcase the abilities of its materials, COPE even built a prototype: the first-ever, completely plastic solar cell.
Prof. Kippelen sees numerous markets for these materials, including organic solid-state devices such as solar cells, OLEDs and field-effect transistors, and noted that the stability of this polymer will ultimately allow manufacturers to use the low-work function electrodes they need for PE systems.
“The new method to produce low-work function electrodes from materials that are stable in air alleviates the use for highly reactive metals, which can pave the way to simplified manufacturing of printed electronic devices in ambient conditions,” Prof. Kippelen said. “The use of air-stable materials that can be printed from solution by all-additive processing has the potential to significantly lower the cost of manufactured printed electronic devices. However, a lot of work remains to be done before this new technology can be transferred into products.”
|
After introducing what appears to be a universal technique to reduce the work function of a conductor in printable electronics, a team led by Georgia Tech's Bernard Kippelen has developed the first completely plastic solar cell. Courtesy: Georgia Institute of Technology. |
One of the key hurdles is utilizing metals in ambient conditions. Conductors such as calcium, magnesium or lithium oxidize when exposed to moisture and oxygen. This requires encapsulation or glass to protect these metals. If a protective polymer can be developed that can be printed in a thin layer, that would go a long way toward solving this issue.
Researchers at Georgia Tech’s Center for Organic Photonics and Electronics (COPE) believe they may have found the answer to this problem. In new findings published in the journal Science, Georgia Tech researchers spread a very thin layer of a commercially available, environmentally friendly polymer, approximately one to 10 nanometers thick, on the conductor’s surface to create a strong surface dipole. The interaction turns air-stable conductors into efficient, low-work function electrodes.
Prof. Bernard Kippelen, director of Georgia Tech’s Center for Organic Photonics and Electronics (COPE), said that the discovery of these stable materials was a result of COPE’s research expertise in numerous fields.
“As often the case in science, the first lead that enabled this discovery was the serendipitous observation that a polymer dispersant that was initially used to help disperse metal oxide nanoparticles was producing unexpected changes of the work function of indium tin oxide electrodes,” Prof. Kippelen said. “A closer look at the chemical structure of this non-conjugated polymer brought us to believe that amine-containing polymers could have some interesting electronic interactions with electrodes that could lead to a reduction of their work function.”
Prof. Kippelen and his team took their findings to their colleagues in Georgia Tech’s Chemistry and Physics departments, who see much potential.
“Sharing of that observation with our closest collaborators in chemistry led to the selection of a few commercially available branched polymers containing simple aliphatic amine groups, including branched polyethyleneimine (PEI) polymers,” said Prof. Kippelen. “The testing of these materials revealed significant reduction of the work function of a variety of conductors. A series of experiments and theoretical modeling with additional collaborators allowed us to get a good physical understanding of the process.
“The validation of the reduction in work function of electrodes in various organic optoelectronic devices exceeded our expectations,” Prof. Kippelen added. “In particular, the good performance of these stable polymers in organic solar cells made us believe that we had made an important discovery that could have a game-changing impact on printed electronics.”
The polymer works with a wide range of metal conductors, including silver, gold and aluminum, as well as graphene and metal oxides. To showcase the abilities of its materials, COPE even built a prototype: the first-ever, completely plastic solar cell.
Prof. Kippelen sees numerous markets for these materials, including organic solid-state devices such as solar cells, OLEDs and field-effect transistors, and noted that the stability of this polymer will ultimately allow manufacturers to use the low-work function electrodes they need for PE systems.
“The new method to produce low-work function electrodes from materials that are stable in air alleviates the use for highly reactive metals, which can pave the way to simplified manufacturing of printed electronic devices in ambient conditions,” Prof. Kippelen said. “The use of air-stable materials that can be printed from solution by all-additive processing has the potential to significantly lower the cost of manufactured printed electronic devices. However, a lot of work remains to be done before this new technology can be transferred into products.”